Benzene Chemical
Price 20.0 INR/ Kilograms
Benzene Chemical Trade Information
- Minimum Order Quantity
- 25 Kilograms
- Payment Terms
- Cash in Advance (CID), Cash Advance (CA)
- Supply Ability
- 25 Kilograms Per Day
- Delivery Time
- 1-3 Days
- Main Export Market(s)
- Australia, Middle East, Western Europe, Central America, Eastern Europe, South America, Asia, North America, Africa
- Main Domestic Market
- All India
About Benzene Chemical
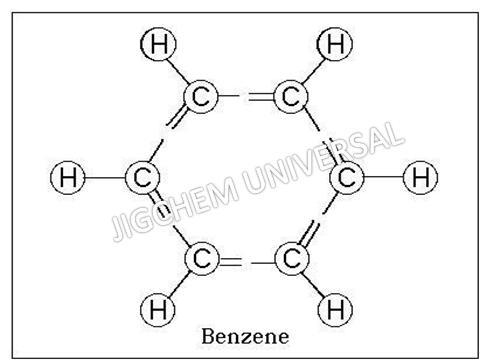
Benzene, or benzol, is an organic chemical compound with the molecular formula C6H6. It is sometimes abbreviated Ph H. Benzene is a colorless and highly flammable liquid with a sweet smell and a relatively high melting point. Because it is a known carcinogen, its use as an additive in gasoline is now limited, but it is an important industrial solvent and precursor in the production of drugs, plastics, synthetic rubber, and dyes. Benzene is a natural constituent of crude oil, and may be synthesized from other compounds present in petroleum. Benzene is an aromatic hydrocarbon and the second annulene 6-annulene a cyclic hydrocarbon with a continuous pi bond.
Uses
In the 19th and early 20th centuries, benzene was used as an after-shave lotion because of its pleasant smell. Prior to the 1920s, benzene was frequently used as an industrial solvent, especially for degreasing metal. As its toxicity became obvious, benzene was supplanted by other solvents, especially toluene (methyl benzene) which has similar physical properties but is not as carcinogenic.

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Veterinary API Category
Robenidine hcl 10% Premix
Price 20 INR / Kilograms
Minimum Order Quantity : 25 Kilograms
Shelf Life : 3 Years
Storage : Dry Place
Physical Form : Powder
Grade : Industrial Grade
Calcium Lactate
Price 20 INR
Minimum Order Quantity : 25 Kilograms
Shelf Life : 3 Years
Storage : Room Temperature
Physical Form : Powder
Grade : Industrial Grade

 Send Inquiry
Send Inquiry






 Send Inquiry
Send Inquiry Send SMS
Send SMS